In high-stakes laboratories, the margin for error is paper-thin.
After all, a single delay or mismatch can compromise patient safety, regulatory compliance, or millions of dollars in R&D.
That’s why more lab leaders are stepping back to reconsider the technology that underpins it all.
Given this, it isn’t surprising that 59% of labs are prioritizing tech upgrades to replace broken or outdated equipment, according to the 2025 State of the Industry Survey by the Medical Laboratory Observer.
In the same survey, 57% of respondents are investing in systems that improve quality while reducing cost.
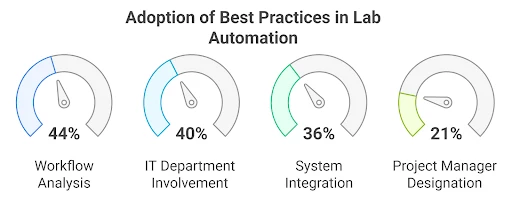
For the sake of convenience, decision makers typically turn to off-the-shelf Laboratory Information Management Systems (LIMS) to replace legacy systems because these are labelled as “good enough” plug-and-play and cost-effective solutions.
Unfortunately, many labs are finding that these introduce just as many problems as they solve.
“Off-the-shelf LIMS systems often fall short in meeting expectations due to lack of flexibility and configurability. In a market of ever-increasing complexity, the need to quickly adapt to changing requirements has never been greater,” said Clynt Taylor, GM of Health Tech and Life Science at Kanda Software.
Custom LIMS are emerging as the smarter alternative — not because they’re flashy or complex, but because they reflect how each lab actually works.
Editor’s Note: This is a sponsored article created in partnership with Kanda Software.
And in a field where precision is everything, that difference matters.
Here’s why more leaders are rethinking “good enough” and going custom instead:
1. Build Compliance Into Everyday Processes
2. Adapt Systems to Lab Growth and Complexity
3. Align Operations with How the Lab Actually Works
4. Protect Sensitive and Proprietary Data
5. Connect Smoothly with All Digital Lab Systems
1. Embed Regulatory Compliance Into the Workflow
Off-the-shelf systems usually treat compliance as add-ons, modules, or fields that help you check boxes and pass inspections.
But that approach assumes regulations are static and one-size-fits-all; something they’re definitely not.
Custom LIMS, such as the ones by Kanda Software, are built around your specific obligations, from CLIA, HIPAA, and FDA 21 CFR Part 11 to ISO/IEC 17025 and everything else in between.
That means fewer surprises during inspections, fewer manual workarounds to meet compliance, and less dependence on your most experienced team members to “remember how things are done.”
Your system itself holds the standard.
2. Scale Systems to Match Lab Growth and Complexity
Off-the-shelf tools are often designed to solve the needs of their time.
Although convenient, this does mean that these systems quickly become outdated when labs add sites, expand services, and install newer technology.
A custom LIMS, on the other hand, is built to evolve.
Modular architecture and integration-ready APIs make it easier to plug into new instrumentation, workflows, or enterprise systems.
This flexibility becomes especially valuable during M&A activity or geographic expansion, where legacy systems often clash.
With a tailored backbone, you can scale without fragmentation.
3. Streamline Operations Around Lab-Specific Workflows
One of the quiet failures of generic LIMS is how they assume all labs function alike. This introduces efficiency-killing issues like:
- Mismatched workflows
- Unnecessary steps
- Manual overrides
On the other hand, custom LIMS are modeled on how your team actually works, from intake and sample prep to data validation and reporting.
That level of alignment shortens onboarding time, reduces training friction, and limits context-switching.
Simply put, you’re not forcing the lab to fit the software. You’re building software that fits the lab.
4. Strengthen Security for Sensitive and Proprietary Data
Labs that work with proprietary research, patient data, or partner IP carry serious security obligations.
Despite the frequency of working with sensitive data, the security of generic systems tends to stop at broad controls like passwords, logins, and antiviruses.
That may be fine for low-risk environments, but in labs handling sensitive IP, genomic data, or patient health information, it’s simply not enough.
Custom LIMS can define who gets access to what — down to individual roles, files, or timestamps.
And real-time audit logs give leadership the visibility they need without slowing anyone down.
When a breach or audit looms, it’s the difference between panic and readiness.
5. Integrate Seamlessly Across the Digital Lab Ecosystem
Labs no longer work in silos, but many of their systems still do.
The average operation now spans a web of instruments, databases, cloud tools, AI models, and compliance software; each with its own language, rules, and blind spots.
By design, custom LIMS can translate, reconcile, and route data across platforms without breaking what already works.
It’s a safeguard against fragmentation, where data goes missing between one handoff and the next.
Likewise, lab leadership doesn’t have to worry about potential compatibility issues, which can be a godsend for labs with specific systems and processes already in place.
Treat Your LIMS Like The Strategic Advantage It Is
Technology choices signal priorities. Opting for the cheapest or fastest implementation may offer temporary relief, but it often sets the stage for long-term inefficiencies.
In high-stakes environments, a lab’s information system shapes how data flows, how decisions get made, and how quickly teams can respond when precision matters most.
That’s why more lab leaders are turning to partners, like Kanda Software, who understand that nuance.
Because in the end, the systems you build today determine how resilient, responsive, and future-ready your lab will be tomorrow.